
"Arm or butt?" my gynecologist barked as she entered the exam room, already tearing the packaging from the prefilled syringe I'd brought to the appointment. I had been trying for almost two hours to have someone, anyone, in the practice stick me with my second dose of the cervical-cancer vaccine Gardasil, to no avail (my doctor had been tied up with an emergency labor; her associate "doesn't do the vaccine"), and I was seriously considering injecting myself.
"Is there a reason your associate wouldn't give me the shot?" I asked, pushing down the waistband of my skirt as the vaccine oozed into my left cheek. "Does she not believe in it? Does she think it's not effective?"
"No, she doesn't give it because she doesn't want to be bothered reminding people to keep up with it," my doctor said, referring to the three doses that must be administered over a six-month period.
"That's why I'm telling you, schedule the third injection now. I'm not going to call you — you're on your own." She scribbled a few notes onto her clipboard and was gone.
My doctor and I had been sparring over Gardasil since I first mentioned it last March. I was 26 and eager for what the American Cancer Society called "one of the most important advances in women's health" — a vaccine against cancer-causing HPV (short for human papillomavirus, the most common STD on the planet). But my physician seemed to view the inoculation as an unnecessary nuisance. Hardly anyone was getting it, she told me, and I of all people didn't need it because I had a long-term boyfriend. "Wait and get it if you break up," she said. "You've already been exposed to any HPV he has by now." When I insisted, she rolled her eyes and handed me a prescription for all three doses. I was to fill it on my own at a specialty pharmacy for $164.95 a pop and bring the needle with me to each of the three injection appointments. "It's an expensive vaccine, so we don't stock it in the office," she explained. She didn't mention that each dose would have to be refrigerated and that I'd soon be triple-wrapping syringes in plastic bags and shoving them to the back of the office Sub-Zero disguised as my lunch.
As I ranted to friends, I learned they were facing similar hurdles. (Even the pharmacist remarked on how many women were filling prescriptions for the vaccine themselves.)
Resistant OB/GYNs rattled off arguments about billing complications, limited safety data, and diminished effectiveness in sexually active women, and many of my peers simply backed off. Sure, I'd read all about the controversy over inoculating girls as young as 9 against an STD. But now the 20-somethings, the women with the highest HPV risk, were being marginalized? It didn't make sense. For me, no wasn't an option, whatever the hassle. I've been terrified of HPV since college, when friends of mine — responsible, condom-using friends — started racking up HPV infections. The scary truth is, more than 80 percent of American women will acquire one by age 50, and the virus is the primary cause of cervical cancer. True, most infections clear up on their own without triggering any symptoms, but the sheer number of people affected and the potential complications had me swearing off sex and referring to men as "carrier monkeys." Research indicating that the virus can be passed through oral sex and heavy petting didn't help. Worst of all, my mother died of cervical cancer when I was 14.
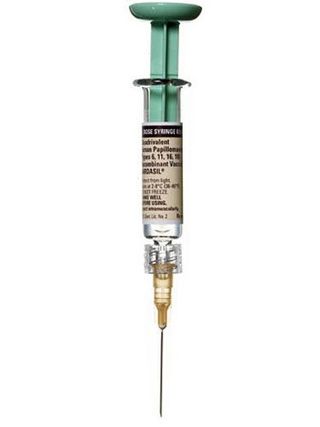
At last, Gardasil promised to quiet the paranoia by protecting against four of the nastiest HPV strains (two responsible for 70 percent of all cervical cancers and two that cause 90 percent of all genital warts) — but there was a catch. The vaccine works best before a person is exposed to the virus, and since most of us will acquire HPV at some point in our lives, that means it works best in virgins. Plus, because the vaccine was originally studied in girls and women ages 9 to 26, the FDA approved it only for that demographic. Many under-26 sex-havers counted themselves out (or were dissuaded by their doctors) — to say nothing of the 27-plus set that was automatically disqualified. I was lucky to still be 26 when I sought out the vaccine — meaning, among other things, that my insurance company would reimburse me the $494.85. But after I completed my last dose, I had lingering questions about the age cutoff and this whole "works best in virgins" business (I know that public-health recommendations need limits, but the "fallen woman" implications stung a bit). So, more than a little frustrated, I called up the experts who have studied HPV for some answers.
Stay In The Know
Marie Claire email subscribers get intel on fashion and beauty trends, hot-off-the-press celebrity news, and more. Sign up here.
For starters, I learned that there's no biological expiration date that magically arrives on a woman's 27th birthday. As Dr. Thomas Herzog of Columbia University Medical Center explained, 26 was the age cap in the original studies because researchers sensibly wanted to focus on the group with the highest rate of HPV infection rather than dilute the findings. What's more, all three doctors I talked to have administered the vaccine to women over 26. "A 27- or 28-year-old who wants to be vaccinated, who understands the limitations of our knowledge in that age group — I'm willing to vaccinate her," added Dr. Kevin Holcomb of Weill-Cornell Medical Center.
In fact, women who have had previous HPV infections (even those that may have developed into cervical precancer) can see some benefit from the vaccine. Holcomb put it this way: "Somebody who's had an abnormal Pap — you don't know exactly which HPV types she's had. I would recommend that you do vaccinate her, with the caveat that she understands it's not going to change the HPV infection she may already have. But it is extremely unlikely that she would have already been exposed to all four types of HPV [prevented by] Gardasil." So much for the fallen-woman disqualifier. Holcomb's approach seemed to back up what I'd felt in my gut all along: My peers and I should do everything we can to protect ourselves (including regular Paps, vaccinated or not), even if we did end up on the tail end of this breakthrough.
Fortunately, the age cap of 26 may soon be a thing of the past. Last November, Merck released data showing Gardasil to be highly effective in women up to 45, which could convince the FDA to extend approval to women over 26. And another version of the vaccine (GlaxoSmithKline's Cervarix, which protects against the same two cancer-causing HPV strains as Gardasil) is expected to win FDA approval early this year, possibly for women as old as 55. Having two versions on the market will most likely bring prices down and eliminate some of the barriers to access — a positive sign that lunch-hour syringe refrigeration and the stick-and-run treatment from reluctant OB/GYNs could be on the way out.
In the meantime, I'm encouraging anyone who will listen to get the vaccine, however infuriating the red tape might be. Because, contrary to what my gynecologist may believe, we don't have to be "on our own" when it comes to HPV protection. As for me, I'm glad I pushed the issue when I did. My boyfriend and I broke up shortly before my last injection.
I would've told my doctor about it, but she was in a hurry.
-
 The Mystery of the Missing Versace Dress Jennifer Garner Famously Wore In ‘13 Going on 30’ Is Fascinating—and Somehow Involves ‘Sex and the City’
The Mystery of the Missing Versace Dress Jennifer Garner Famously Wore In ‘13 Going on 30’ Is Fascinating—and Somehow Involves ‘Sex and the City’Every dress has a story, and this one is complex.
By Rachel Burchfield Published
-
 Selena Gomez References Her Own Character on Date Night
Selena Gomez References Her Own Character on Date NightThe singer attended the Knicks game in an 'Only Murders'-coded coat.
By Julia Gray Published
-
 Ashley Judd Says an “Undiagnosed” Mental Illness “Stole” Her Mom Naomi Judd
Ashley Judd Says an “Undiagnosed” Mental Illness “Stole” Her Mom Naomi Judd“I carry a message of hope and recovery.”
By Danielle Campoamor Published
-
 Senator Klobuchar: "Early Detection Saves Lives. It Saved Mine"
Senator Klobuchar: "Early Detection Saves Lives. It Saved Mine"Senator and breast cancer survivor Amy Klobuchar is encouraging women not to put off preventative care any longer.
By Senator Amy Klobuchar Published
-
 How Being a Plus-Size Nude Model Made Me Finally Love My Body
How Being a Plus-Size Nude Model Made Me Finally Love My BodyI'm plus size, but after I decided to pose nude for photos, I suddenly felt more body positive.
By Kelly Burch Published
-
 I'm an Egg Donor. Why Was It So Difficult for Me to Tell People That?
I'm an Egg Donor. Why Was It So Difficult for Me to Tell People That?Much like abortion, surrogacy, and IVF, becoming an egg donor was a reproductive choice that felt unfit for society’s standards of womanhood.
By Lauryn Chamberlain Published
-
 The 20 Best Probiotics to Keep Your Gut in Check
The 20 Best Probiotics to Keep Your Gut in CheckGut health = wealth.
By Julia Marzovilla Published
-
 Simone Biles Is Out of the Team Final at the Tokyo Olympics
Simone Biles Is Out of the Team Final at the Tokyo OlympicsShe withdrew from the event due to a medical issue, according to USA Gymnastics.
By Rachel Epstein Published
-
 The Truth About Thigh Gaps
The Truth About Thigh GapsWe're going to need you to stop right there.
By Kenny Thapoung Published
-
 3 Women On What It’s Like Living With An “Invisible” Condition
3 Women On What It’s Like Living With An “Invisible” ConditionDespite having no outward signs, they can be brutal on the body and the mind. Here’s how each woman deals with having illnesses others often don’t understand.
By Emily Shiffer Published
-
 The High Price of Living With Chronic Pain
The High Price of Living With Chronic PainThree women open up about how their conditions impact their bodies—and their wallets.
By Alice Oglethorpe Published