Why I Chose to Get the COVID-19 Vaccine at 32 Weeks Pregnant
Like many pregnant women—and many Black Americans—Dr. Toluwalase Ajayi was apprehensive about getting the vaccine at first.

Toluwalase Ajayi, M.D., is a pediatrician, palliative physician, and clinical researcher in her late 30s living in San Diego, California. On January 9, at 32 weeks pregnant, Dr. Ajayi got the COVID-19 vaccine, despite initial concerns about the lack of clinical trials on pregnant women, and the rightful distrust many people in the Black community have about the health care system. Here, she tells Marie Claire, in her own words, how she came about her decision and why she'll continue to advocate for the inclusion of underrepresented populations in research studies.
The vaccine became available to us [at both hospitals where I work] in late December. Early on in my pregnancy, my plan was to wait to get the vaccine until after I gave birth, but I am exposed by my jobs, so I decided to take my own medical advice. I talked to my OB-GYN about it. She said, You're high risk, you should actually get it. You've always done well with vaccines, you've never had any issues. She suggested I wait until I have a little bit of a break in my clinical schedule to get the vaccine. I was working pretty much through Thanksgiving, Christmas, and New Year's, so I didn't get that break in time until I was 32 weeks pregnant.
I was mainly concerned about the fact that there weren't pregnant women enrolled in the vaccine trials. So we just didn't know conclusively, like we did with other patient populations, the effect on the mom and the effect on the fetus. Regardless of whether or not pregnant women had been included in the clinical trials, there was a disproportionately low amount of minorities who participated in the trials—[at least in part] because there's a lot of fear around and a lot of historical racism that happened with clinical research. It's a vicious cycle. We don't participate because of what happened to us in the past, but because we don't participate, we don't get the data that we need to see to feel comfortable.
I also have pregnancy-induced thrombocytopenia. One of the rare side effects of the vaccine is an immune reaction that can trigger thrombocytopenia and potentially drop my [platelet] counts further. That was the one piece of information that really, really frightened me. So I talked to my hematologist friend and my OB-GYN friend, and they're like, "Listen, the risk of that exists with any vaccine. It's actually higher for the MMR (measles, mumps, and rubella) [vaccine] than it is for the COVID vaccine." So once I got that piece of information, I was happy with it. My husband said, "Do whatever you feel comfortable with and I support you either way." My sisters and my mom were very nervous for me. I actually didn't tell them that I went ahead and decided to do it. They saw [my post] on Twitter. My mom can be very persuasive, but I was like, I'm just going to do it and then we'll talk about it afterwards.
It's a vicious cycle. We don't participate because of what happened to us in the past, but because we don't participate, we don't get the data that we need to see to feel comfortable.
I got the vaccine at Scripps Green Hospital here in La Jolla, where I work. There was a small group of us pregnant Black women who talked about getting it at the same time. One of them said, "You know, I'm going to wait." Another one of them was like, "I'm past the first trimester. I'm doing it."
I had the option of getting either the Moderna or the Pfizer vaccine—I didn't care which one—and I ended up getting Pfizer. It was really painless. It hurt less than a flu shot and a whole lot less than the tetanus shot did. It was funny because when I walked in, everybody was looking at me because I'm very pregnant and I don't think they had vaccinated anybody else who was visibly pregnant yet. I asked the nurse and she was like, "Yep, you are my first pregnant woman that I'm vaccinating." She said, "Hey, when you get it, just kind of move your arm around a little bit so it's not too sore." So I did that. Then after the shot, they had me wait. Normally they have you wait 15 minutes afterwards, but she had me wait five extra minutes to make sure that I wasn't having any reactions to it. Then I went to my clinical shift at work.
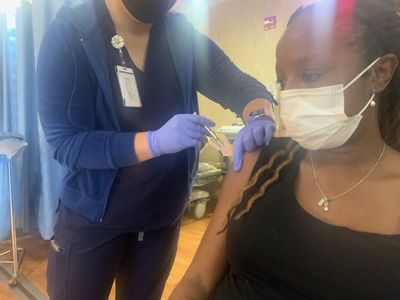
Dr. Ajayi receiving the Pfizer vaccine on January 9, 2021.
My arm was a little bit sore, but by the end of the day, the soreness was gone. What I felt more was the increased fatigue. The next morning, I woke up exhausted and I had a small headache. That could have been due to dehydration, the fact that I was finishing nine days in a row in the hospital, all that. My blood pressure was also a little bit up before I went to work the next day, but by the time I got home from work, my blood pressure was back to normal. My headache was gone. I didn't even take any Tylenol.
Get exclusive access to fashion and beauty trends, hot-off-the-press celebrity news, and more.
My second vaccine is scheduled for Saturday, January 30.
I decided to post a tweet about receiving the vaccine. It was definitely spur of the moment, but I was like, You know what, if I'm going to do this and I'm going to tell my patients to do this, I'm going to be public and share what it's like going through that journey. I was sitting in the chair, like when they're observing you, and having people walk by and look at me and I was like, I'm just going to do it. It wasn't anything professional. I just took a selfie and said, "I'm doing this and I'm sharing this because I was also one of those Black women who was nervous about getting the vaccine, but I'm doing it because I know it's better than getting the disease."
As an adult palliative care physician, I see way too many Hispanic and Black patients in the ICU whom I do compassionate extubation for, where we remove them from life support. It's devastating. This community is so family oriented—family and being together is such a big part of our culture. To have that happen and not have your family be able to say goodbye to you, to not have your family be able to come visit you because we don't allow visitors in the hospital when you're COVID positive? It's just devastating. I was probably thinking about that as well [when I decided to get the vaccine].
I know a lot of pregnant women and people of color are nervous about getting the #CovidVaccine. I am 32 weeks and 1 day today and just received the @pfizer vaccine. I feel like #ThisIsOurShot to protect us and our babies. I am a #PowerMom and will post how I feel how I react so pic.twitter.com/bJajEWtowRJanuary 9, 2021
After getting the vaccine, there were a couple of research trials that I joined to report on how I felt. It's part of the work I do at Scripps Research. I have an app called PowerMom, and the goal is to collect information about what happens to women during their pregnancy. It's because, in general, pregnant women are left out of clinical research because we're seen as a vulnerable population. As a result, we don't get to know a lot of things about our body and what's happening to us. So, PowerMom really is to get all of that information and then feed that information back to the women, so you see that you're not alone. I will also be collecting data from pregnant woman who received the vaccine, and tracking their symptoms to get a large database of that as well. The relaunch [of the app] will focus on COVID, the vaccine and symptoms, and really target underrepresented minorities.
I would love it if the people who are conducting the research studies looked more like the people we are trying to recruit and create more funding opportunities for. I would also like us to really look at what barriers exist in enrolling Black women in medicine, trials, and research, because a lot of times you don't take into account transportation and childcare. We don't consider the things that make it hard to be a Black woman in general because those are not the people who are doing the studies. I would love to see the government be more proactive about that. And, honestly, that goes with any disproportionate population.
#ThisIsOurShot update: Happy #MLKDay2021 ! Today I am 33 and 3 days pregnant, and had my OB visit today; 9 days after my first #COVID19 @pfizer vaccine. Baby is growing and developing well. Fluid level is normal and baby is measuring at 34/35 weeks, which is how I feel. pic.twitter.com/APpfnEkrPUJanuary 19, 2021
I don't blame [pregnant Black women] for being hesitant about receiving the vaccine because we don't have good information out there about the vaccine. What we do have is solid information that if you're Black, if you're Hispanic, you're more at risk of dying [from COVID-19]. You're more at risk of getting admitted to the ICU. We know that the disease is worse than the vaccine. And because we're such a high-risk population, we also know that pregnant women do worse with COVID-19. So, consider the fact that you're an underrepresented minority and you're pregnant. You don't want to take that chance. The known facts of the disease are greater than the uncertainty about the vaccine.
If you're pregnant, the CDC's advisory committee recommends talking to your doctor about receiving the COVID-19 vaccine. To learn more about the COVID-19 vaccine, including safety and administration protocols, click here.
RELATED STORIES


Rachel Epstein is a writer, editor, and content strategist based in New York City. Most recently, she was the Managing Editor at Coveteur, where she oversaw the site’s day-to-day editorial operations. Previously, she was an editor at Marie Claire, where she wrote and edited culture, politics, and lifestyle stories ranging from op-eds to profiles to ambitious packages. She also launched and managed the site’s virtual book club, #ReadWithMC. Offline, she’s likely watching a Heat game or finding a new coffee shop.

